I got an invitation from ….
-
From his lips to gods ears.
Of course there’s such a thing as ‘too good’ - for example, they could say that they’ll give conditional approval for rare disease treatments based on safety alone and let efficacy be proven in a P4 (post marketing) study. That sounds awesome, but the reality is insurance companies wouldn’t approve it considering it ‘experimental’.
What I’d like to see is them allow more flexibility with approvals based on biomarkers rather than clinical outcomes which can take much more time to demonstrate in some diseases.
-
@jon-nyc said in I got an invitation from ….:
So my flights are canceled. I guess I’m going to dial in.
When I’m not shoveling.
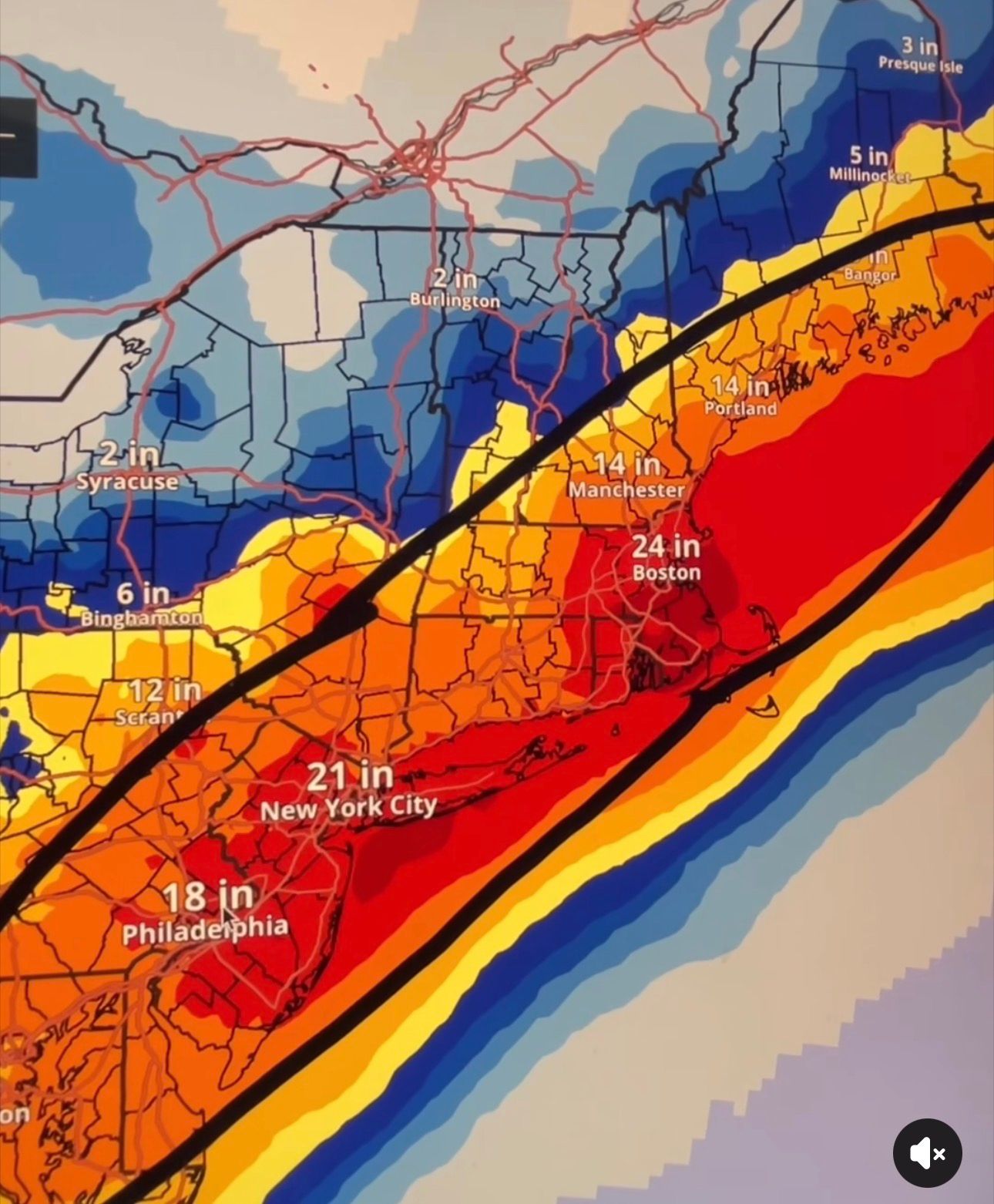
Snorting Coke of your own toilet just isn’t the same.
-
The plausible mechanism pathway. That would be awesome for us but it’s only for ultra-rare, which has no formal definition but probably would exclude us.
Hello! It looks like you're interested in this conversation, but you don't have an account yet.
Getting fed up of having to scroll through the same posts each visit? When you register for an account, you'll always come back to exactly where you were before, and choose to be notified of new replies (either via email, or push notification). You'll also be able to save bookmarks and upvote posts to show your appreciation to other community members.
With your input, this post could be even better 💗
Register Login