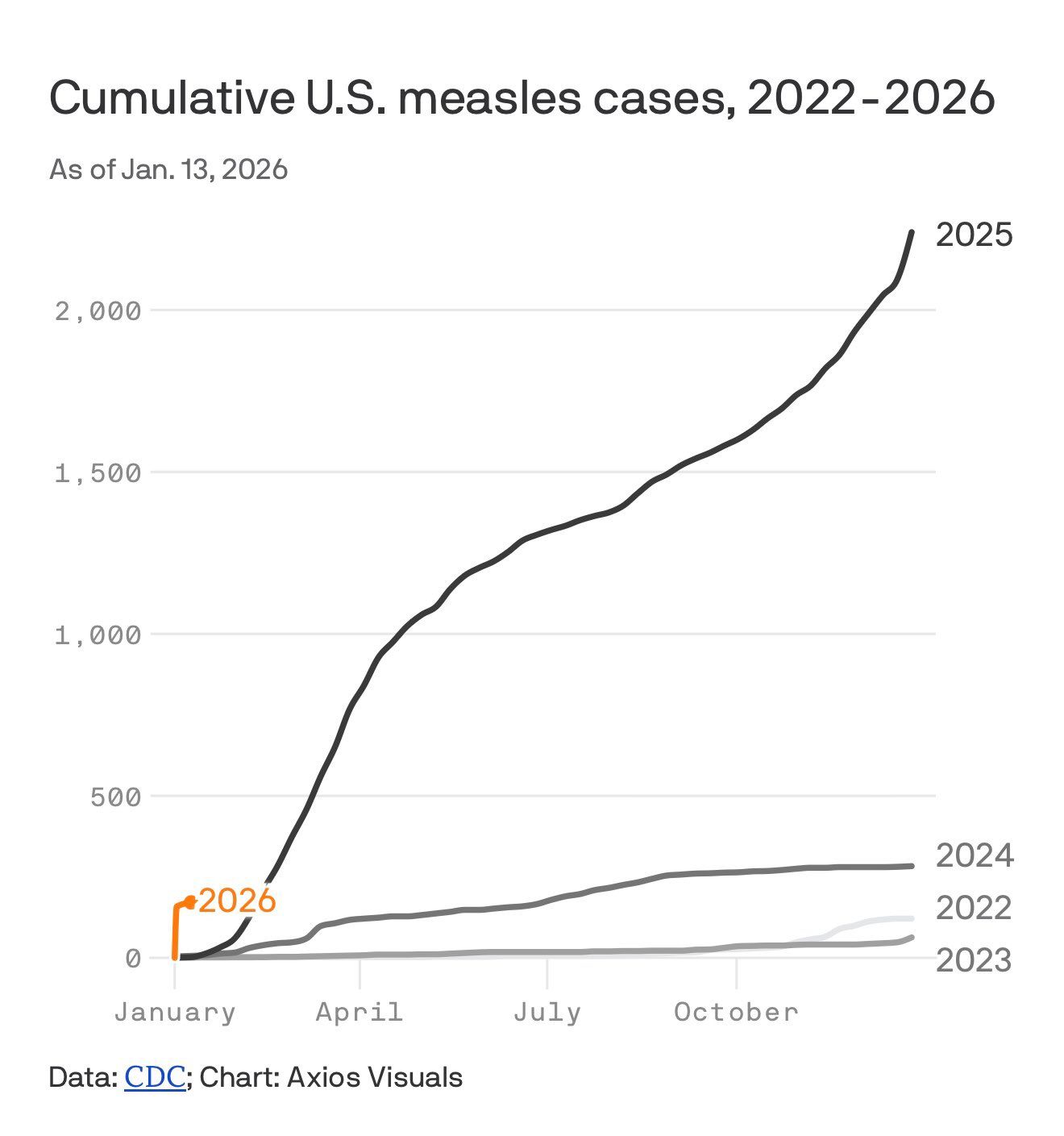
Beyond the antivaxx stuff MAHA is either banal or fake
-
"I'm not afraid of children dying, I used to sell them drugs"
-
Could easily lay some blame at the feet of the self-righteous covid hawks who couldn't get enough of jamming vaccines down everybody's throat. Like it or not, this is part of the backlash, and it's hardly obvious that any of that was a good idea, now or at the time. To whatever extent this is even a blameworthy event. Not everybody is hyper focused on private industry finding profitable cures for rare diseases.
-
It's not quite that stark an about face. The refusal letter prominently encouraged Moderna to come and have a sit down with the FDA to resolve the issue. Maybe that happened. "Massive pushback caused the FDA to relent" is a crafted narrative, unless more evidence was provided in the paywall section.
-
Zero chance they had a their type A meeting in the time since this unfolded. Far more lijkely is Senators applied pressure (ie, the system worked) and FDA got some face saving vague promise for a post marketing study.
said in Beyond the antivaxx stuff MAHA is either banal or fake:
Far more lijkely is Senators applied pressure ...
And administration friendly investors. (A16Z has a biotech fund after all). What was particularly insidious about this decision was indication that the FDA couldn’t be trusted to keep to its own written word. How are you supposed to invest hundreds of millions into a p3 trial when the FDA backtracks on its own written approval of the design after the funds have been spent?
-
Anyway, the official story is that Moderna made changes to their application, and the FDA accepted the revised version. Everything else is narrative. But the implication that the FDA simply reversed is not accurate. Moderna did make changes to the app. According to my exhaustive google search.
-
You seem confident that the FDA can reverse a written decision on the spot, but are unable to expedite a meeting on the spot.
@Horace said in Beyond the antivaxx stuff MAHA is either banal or fake:
You seem confident that the FDA can reverse a written decision on the spot, but are unable to expedite a meeting on the spot.
Meetings take a lot of preparation, presentations, data, etc. Writing a letter agreeing to review a submission in the future is trivial and was clearly done regardless of what else happened.
Hello! It looks like you're interested in this conversation, but you don't have an account yet.
Getting fed up of having to scroll through the same posts each visit? When you register for an account, you'll always come back to exactly where you were before, and choose to be notified of new replies (either via email, or push notification). You'll also be able to save bookmarks and upvote posts to show your appreciation to other community members.
With your input, this post could be even better 💗
Register Login