Chinese Syringes
-
FDA Issues a Critical Safety Communication on Syringes Manufactured in China
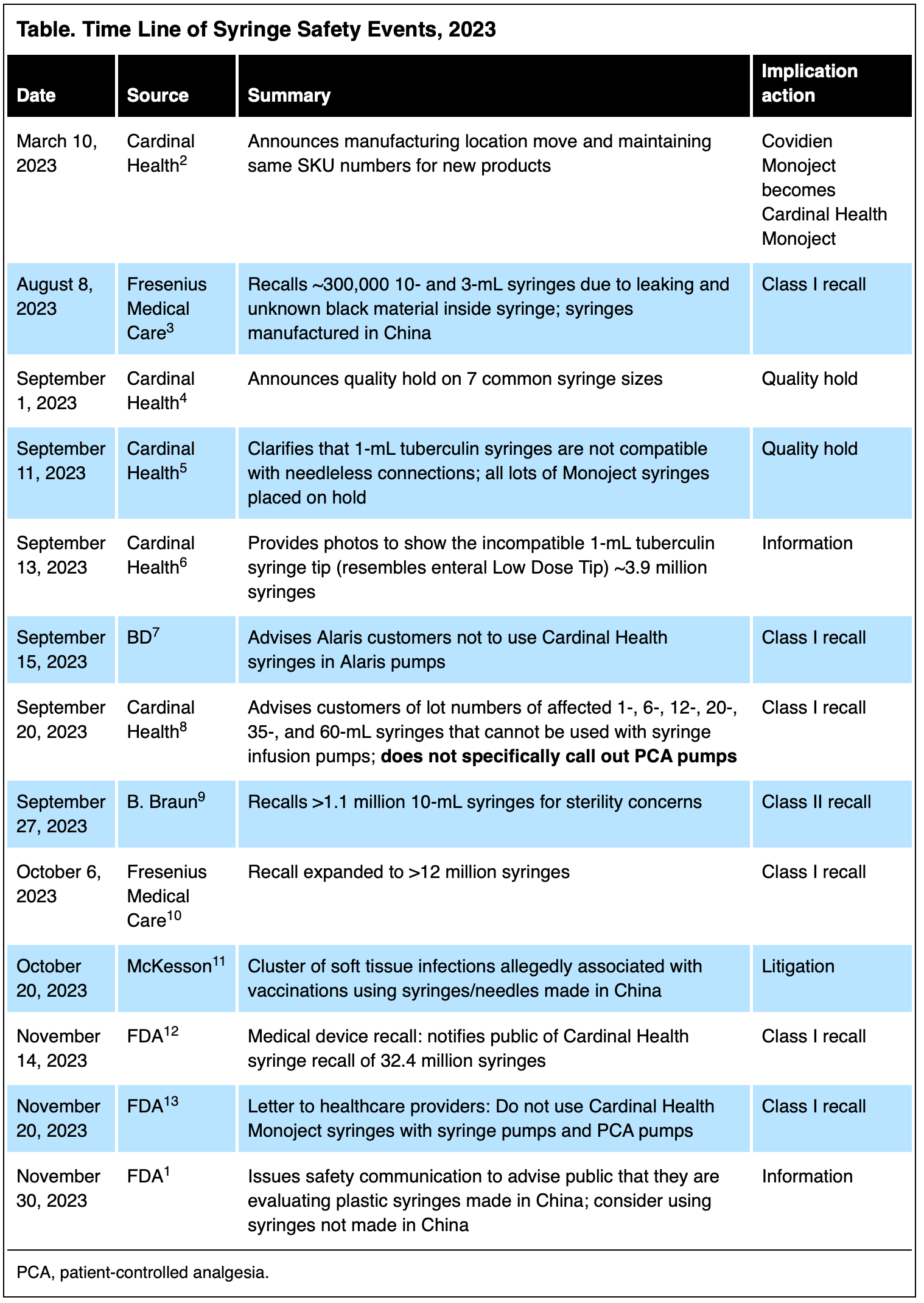
A series of events could lead up to a supply chain disruption of one of our most critical medical devices. On November 30, 2023, the FDA issued a safety communication alerting consumers, healthcare providers, and medical facilities regarding an ongoing evaluation of potential device malfunctions linked to syringes manufactured in China and sold under familiar American names.1
The FDA has received reports detailing quality concerns related to multiple Chinese syringe manufacturers. Issues reported include leaks, breakage, and additional issues due to manufacturer changes made to the syringe dimensions. These quality issues may affect the performance and safety of the syringes, including their ability to deliver the correct dose of medication when used alone or with other medical devices such as infusion pumps.
The agency is expressing apprehension that specific syringes originating from China might lack uniformity and fail to meet the required standards of quality and performance. Currently glass syringes, oral syringes, and syringes manufactured in the United States are not affected by this communication.
Of note, a majority of US brands source their syringes from Chinese manufacturers. At this time Becton, Dickinson and Company (BD) is the only domestic syringe manufacturer that sells under its own label. The current recalls cover more than 45 million disposable syringes in the US market. In the November 30, 2023, communication, the FDA stated it will continue to monitor for additional quality and safety concerns and may prevent further shipments of Chinese-made syringes from entering the United States. The affected supply of syringes, in addition to continued diligence by the FDA to monitor ongoing supplies from China, may lead to shortages of certain sizes to all syringes and increase in costs.
Before the FDA’s communication, US medical device manufacturers of syringes had cautioned about compatibility problems between their syringe with syringe infusion pumps and other quality problems. The time line in the Table highlights events leading up to the November 30 FDA communication.
Standards for Syringes
Syringes are often viewed as an interchangeable commodity with little to no concern for variations. The FDA “clears” (approves for marketing) syringes for use in the United States under the 510(k) device process. The FDA utilizes a 3-class system to classify medical devices based on risk, where Class I devices (ie, bandages) have the lowest risk to patients and Class III devices represent the highest risk category (ie, pacemakers, implants). Syringes are classified as a Class II medical device. Class II medical devices carry a moderate to high risk to the patient and/or user.
The International Organization for Standardization (ISO) sets global standards for syringes, ensuring their quality, safety, and performance. ISO has established essential standards for syringes:
ISO 7886-1:2017 Sterile hypodermic syringes for single use — Part 1: Syringes for manual use applies to syringes that healthcare professionals manually operate, and ISO 7886-2:2020 Sterile hypodermic syringes for single use Part 2: Syringes for use with power-driven syringe pumps focuses on syringes designed for compatibility with automated medical devices. These guidelines are in place to ensure that syringes meet the following criteria:
- manufactured using tested and approved materials that meet strict safety standards;
- contain accurate graduations for dosing (with allowance for accuracy variances);
- effectively deliver medications;
- maintain sterility; and
- reliably and consistently function as designed.
Syringes that vary in calibration and sizes pose additional challenges to healthcare providers, as they relate to dose accuracy. Some syringes have different barrel markings, 0.1 mL versus 0.2 mL, which can lead to dosing errors if syringe brands are switched, or manufacturers of syringes make changes, and the healthcare provider is not apprised of the change.
Syringes that vary in physical size but deliver the same quantity create issues with syringe pumps and robotic compounding devices that have been calibrated to a specific size. The dimensional variances may result in syringe pump issues leading to potential underdosing, overdosing, occlusion alarms, and potential delays in therapies. The FDA highlighted the dimensional variances in the November 14, 2023, FDA medical device recall: Cardinal Health recalls Monoject disposable syringes for incompatibilities with syringe pumps.12 The FDA posted a picture of dimensional variances of Monoject 10-mL syringes that deliver the same volume. The Figure shows an example of dimensional differences between 2 syringes, both named “Monoject.” Once the syringe is removed from the package, the distinction between Covidien and Cardinal Health brands is gone. Syringe and PCA pumps have libraries of validated syringes, often labeled simply “Monoject.” This has caused a Class I recall/product correction for Alaris PCA and syringe pump users to update their compatibility lists.14
The current situation may be further complicated if the FDA chooses to temporarily prohibit importation of syringes from China. Hospitals that prepare and administer antibiotics as IV push should consider using a mini-bag system as an alternative to conserve 10-mL syringes. If you purchase medications that are prepared by commercial compounding companies, consult with the company to ensure that they are not using syringes from China, or if they have recently switched away from China syringes, that drug stability testing has been done with the new brand of syringes.
-
Hadn't noticed any problems, but we have BD syringes. Sad about Monoject. I used to prefer Monoject over BD, because while BD made a good 10ml syringe (the most common size I use), Monoject made a 12 ml that was just a hair bigger. Sometimes a couple of mls makes a difference.
Hello! It looks like you're interested in this conversation, but you don't have an account yet.
Getting fed up of having to scroll through the same posts each visit? When you register for an account, you'll always come back to exactly where you were before, and choose to be notified of new replies (either via email, or push notification). You'll also be able to save bookmarks and upvote posts to show your appreciation to other community members.
With your input, this post could be even better 💗
Register Login